
Late 2020.
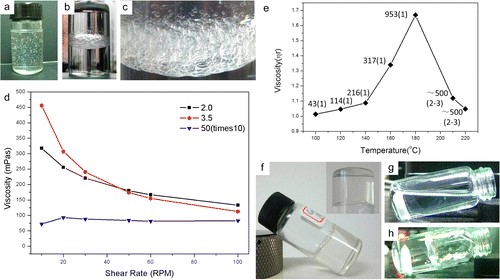
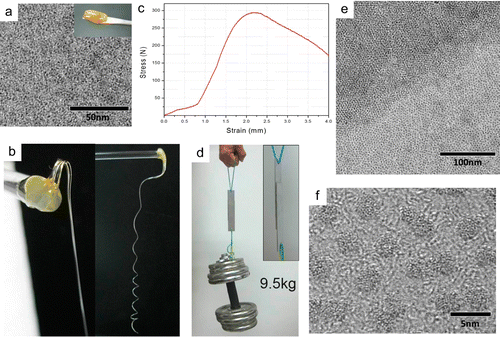
As building materials, inorganic nanocrystals can be difficult to work with. Conventional nanoparticles and nanowires are often rigid and brittle, and require careful coaxing to assemble into complex structures. If only inorganic nanomaterials were more like flexible organic proteins and other macromolecules, researchers could create much more sophisticated shapes for novel devices.Now with a simple solution-based method, we have created a series of sub-nanometer materials, like GdOOH ultrathin nanowires, indium sulfide nanocoils, etc. They’re so thin and long that they bend like flexible proteins or polymers.We suggest that these ultrathin structures may help researchers to bridge the gap between organic and inorganic nanomaterials, providing insights into superstructure self-assembly and polymer crystallization..
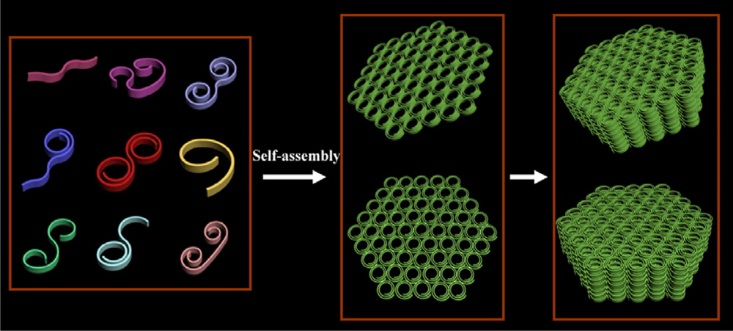
Size may determine many properties in nanoscale. Here we are interested in building nanostructures from the self-assembly of ultrathin sub-units. There are many kinds of driving forces in the field of self-assembly, and each of them may take control under suitable situation. Recently we found that when the size of the inorganics decrease to the molecular scale, weak interactions would dominate the interplay between sub-units and environments. In light of this, “rigid” inorganic could achieve previously unnoticed flexibilities in proper condition, like organic molecules. Consequently, more complicated morphologies could be achieved and foreseen. Self-assembly hold great promise for constructing sophisticated nanostructures, and by utilizing the weak interactions between ultrathin sub-units, we may be able to construct nanostructures in the way like building molecules in organic chemistry..